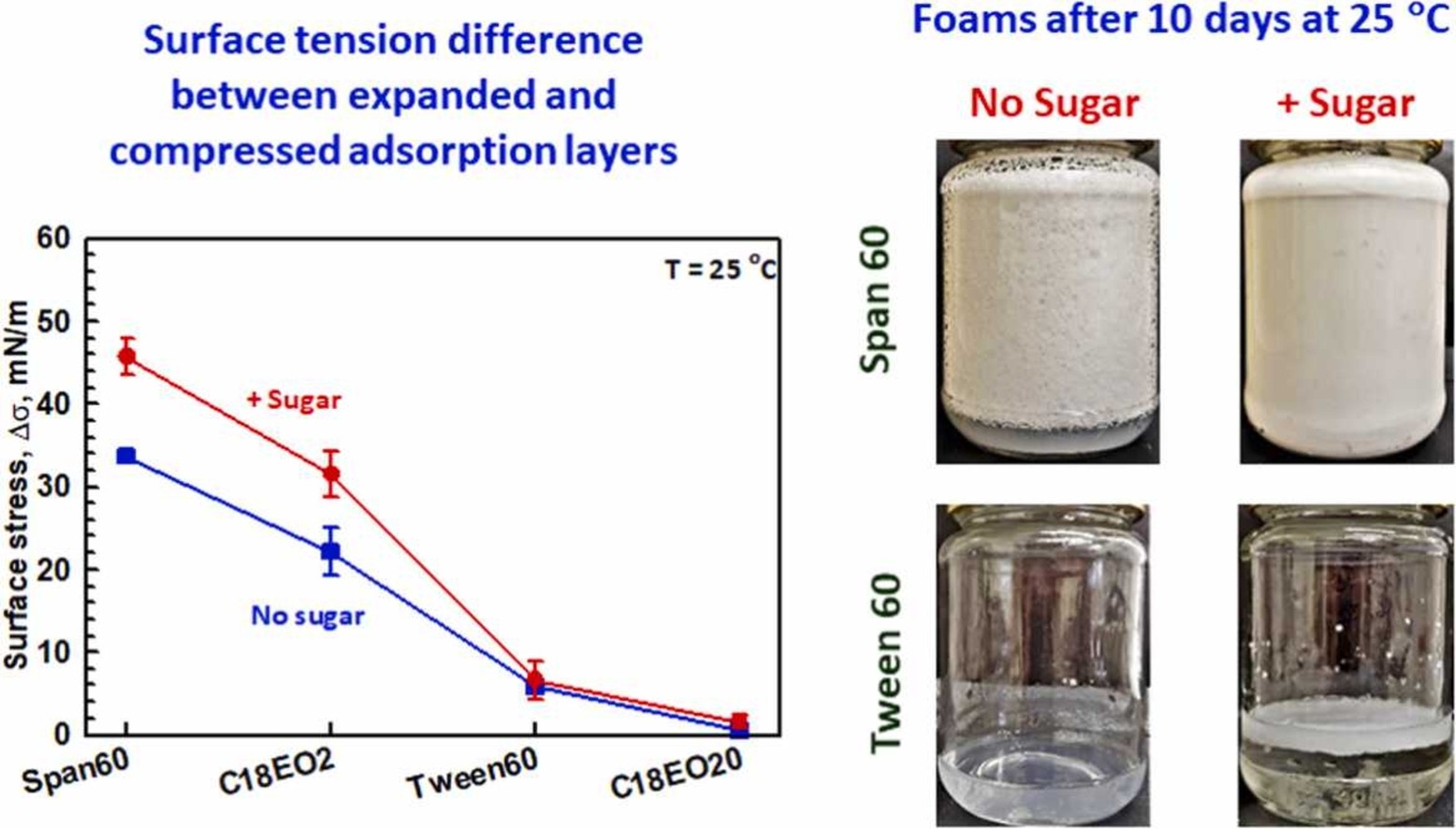
The surface and foam properties of two oil-soluble surfactants, Span 60 and Brij 72, and two water-soluble surfactants, Tween 60 and Brij S20, are compared. Aqueous surfactant solutions containing sugar at high concentration (63 wt%) are also studied in the context of sweet food-related foams. The experimental results show that foams with very small bubbles of ca. 5 µm are formed and remain stable for more than 10 days at room temperature when oil-soluble surfactants are dispersed in the sugar-rich solutions. The excellent stability of these foams is related to (1) The multilamellar vesicles present in the respective surfactant dispersions which increase the viscosity and decrease the rate of water drainage from the foam; (2) The very slow exchange of surfactant molecules between the interface and the adjacent aqueous solution that leads to very significant difference in the surface tension of shrinking and expanding bubbles, which in turn decreases the rate of bubble Ostwald ripening. The foams formed from Span 60 are more stable as compared to the foams formed from Brij 72, due to the higher viscosity and the slower interface-solution exchange. The foams formed from Brij S20 solutions are least stable - they are completely destroyed after 1 day at room temperature, even in the presence of sugar. The main conclusion of this study is that oil soluble surfactants can form condense adsorption layers on the bubble surfaces after adsorption from aqueous solutions with and without sugar in it. The formed layers, which present a better molecular packing, reduce the gas permeability and decelerate the Ostwald ripening disproportionation of the foam This is obtained by a relatively low interfacial tension imparted by continuous shrinkage and expansion of bubbles during foam generation, ultimately ensuring long-term stabilization of formed foams.