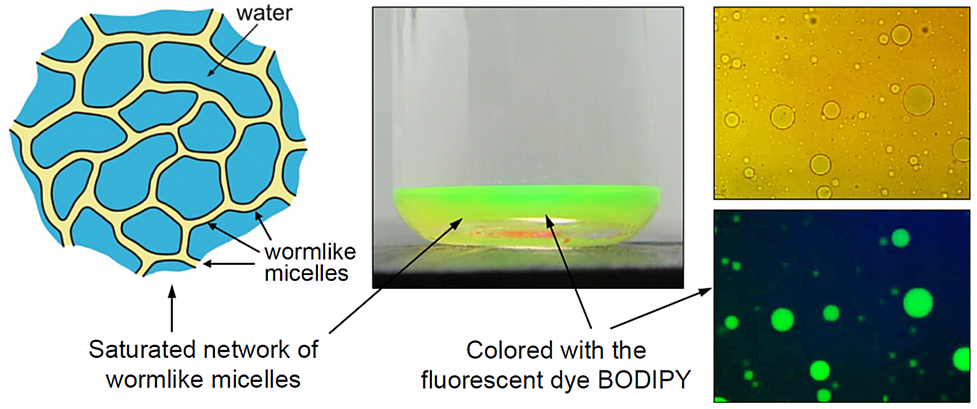
Phase separation of saturated micellar network, as a result of cross-linking of branched micelles, is established in mixed solutions of the anionic surfactant sodium laurylethersulfate (SLES) and the zwitterionic cocamidopropyl betaine (CAPB) in the presence of divalent counterions: Ca2+ , Zn2+ and Mg2+. The saturated network appears in the form of droplets, which are heavier than water and sediment at the bottom of the vessel. In the case of Mg2+, the sedimented drops coalesce and form a separate multiconnected micellar phase – a supergiant surfactant micelle. For this phase, the rheological flow curves show Newtonian and shear-thinning regions. The appearance/disappearance of the Newtonian region marks the onset of formation of saturated network. The addition of small organic molecules (fragrances) to the multiconnected micellar phase leads to an almost spontaneous formation of oil-in-water nanoemulsion. The nanoemulsification capacity of the multiconnected micellar phase decreases with the rise of the volume of the oil molecule. A possible role of the network junctions in the nanoemulsification process can be anticipated. The properties of the multiconnected micellar phases could find applications in extraction and separation processes, in drug/active delivery, and for nanoemulsification at minimal energy input.