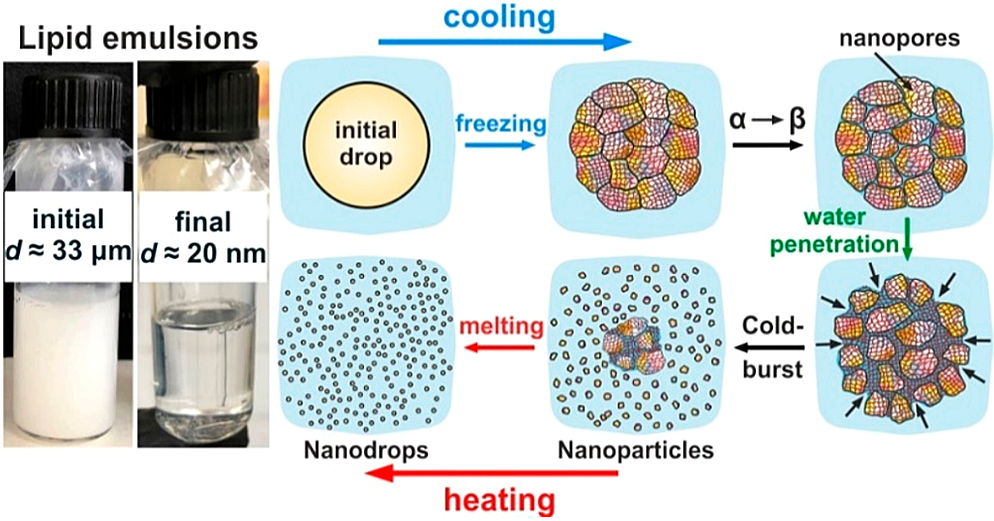
We describe several unexpected phenomena, caused by a solid−solid phase transition (gel-to-crystal) typical for all main classes of lipid substances: phospholipids, triglycerides, diglycerides, alkanes, etc. We discovered that this transition leads to spontaneous formation of a network of nanopores, spreading across the entire lipid structure. These nanopores are spontaneously impregnated (flooded) by water when appropriate surfactants are present, thus fracturing the lipid structure at a nanoscale. As a result, spontaneous disintegration of the lipid into nanoparticles or formation of double emulsions is observed, just by cooling and heating of an initial coarse lipid-in-water dispersion around the lipid melting temperature. The process of nanoparticle formation is effective even after incorporation of medical drugs of high load, up to 50% in the lipid phase. The role of the main governing factors is clarified, the procedure is optimized, and the possibility for its scaling-up to industrially relevant amounts is demonstrated.